Get in touch with our team of experts:
Introducing Cryoport Systems’ IntegriCell® Cryopreservation Services
Where cryopreservation meets the power of our end-to-end supply chain platform, revolutionizing cell therapy logistics with integrated solutions.
Our IntegriCell® cryopreservation service offering provides an integrated, end-to-end solution that ensures the highest quality, consistency, and viability of manufacture-ready cryopreserved leukopaks for advanced cell therapies. Cryoport Systems combines state-of-the-art cryopreservation services with a robust, global supply chain platform and a track record of success that spans decades. Our IntegriCell® offering enables seamless, efficient, and reliable cryopreservation combined with end-to-end supply chain services for critical cellular materials, ultimately supporting the successful treatment of patients worldwide.
- Standardized Protocols
Our rigorous protocols developed by seasoned experts are designed to meet the highest quality standards and ensure consistency across our global facilities. - Optimized Cellular Quality
IntegriCell® cryopreservation services are designed to optimize cellular quality through processing within an ideal 24-hour window (post-collection) to maximize viability. - Global Reach
With facilities strategically located in the U.S. and Europe, we are able to support your projects worldwide. - Integrated Services
Cryopreservation, biostorage, consulting, and logistics are seamlessly integrated into our end-to-end platform for streamlined operations and maximum efficiency.
Unlock the Full Potential of Advanced Cell Therapies with IntegriCell™ Cryopreservation Services
Redefining the Standards for Leukapheresis-derived Cell Therapies with IntegriCell® Cryopreservation
Ensuring superior quality and consistency in manufacture-ready cryopreserved leukopaks for advanced cell therapies.
IntegriCell® cryopreservation services from Cryoport Systems redefine the standards for cell therapy by enhancing the safety, quality, and viability of manufacture-ready cryopreserved leukopaks. Our state-of-the-art cryopreservation process, integrated with our global temperature-controlled supply chain platform, provides unmatched reliability, efficiency, and scope. From cryopreservation to final delivery, we ensure every step is meticulously managed, empowering advanced cell therapies to reach patients with unparalleled consistency and support.
Cellular Viability and Process Optimization
IntegriCell® cryopreservation services are designed to maximize cellular viability and optimize the cryopreservation process. Our state-of-the-art cryopreservation protocols ensure that cellular quality is preserved within the critical 24-hour window (post-collection), maintaining the integrity and effectiveness of manufacture-ready cryopreserved leukapheresis material. By integrating our standardized cryopreservation processes with robust logistics management, we ensure the safe and secure delivery of high-quality cryopreserved leukopaks.
- Closed Automation Process: Our GMP-compliant, closed automated process significantly reduces contamination risk and associated quality variation from human intervention.
- Extended Shelf Life: Cryopreservation within 24 hours (post-collection) can preserve the quality of starting material to uncouple the collection of starting material from drug product production and manufacturing.
Standardization for Consistent Quality
Standardized protocols across all IntegriCell® services ensure the consistency and quality of manufacture-ready cryopreserved leukopaks across our global network. Our experienced team adheres to strict compliance standards, ensuring uniformity and reliability in every cryopreservation process.
- Experienced Team: Over 25 years of cryopreservation experience.
- Global Standardization: Uniform standards across all Cryoport Systems locations and facilities.
- Reduced Variability: Through the application of consistent cryopreservation processes.
Maximizing Manufacturing Efficiency
IntegriCell® cryopreservation services enhance manufacturing slot utilization to drive cost savings and maximizing product revenue. By providing manufacture-ready cryopreserved leukopaks, we offer therapy developers flexibility and reliability in their production schedules.
- Scheduling Flexibility: Cryopreservation allows for manufacturing initiation up to seven days a week.
- Cost Control: Scheduling flexibility alleviates the reactive time sensitivity of working with fresh, donor-derived cellar materials for improved efficiency.
- Reduced Backlogs: Decreases logistical challenges and enhances manufacturing success rates.
Global Coverage & Support
Cryoport Systems’ IntegriCell® cryopreservation services cover a global scale, supporting clinical trials by ensuring the preservation and delivery of high-quality manufacture-ready cryopreserved leukopaks. Our expansive network of facilities enables broader access to patient pools, which is crucial for the development of cell and gene therapies.
- Geographic Coverage: Cryopreservation facilities in the U.S. and Europe for worldwide reach.
- Expanding Footprint: Increasing access to patients and donors outside metropolitan areas through our Global Supply Chain Centers.
- Global Logistics: Supported by our network of global facilities, allowing the transport of sensitive materials across the U.S., European, and APAC regions.
One Supply Chain Service Provider
Partnering with Cryoport Systems for your cryopreservation needs means leveraging our integrated supply chain solutions to optimize costs, improve efficiencies, and mitigate risks. From cryopreservation to final drug product, our end-to-end services provide a seamless experience for therapy developers and are all managed within a single vendor relationship.
- Integrated Services: Cryopreservation, storage, logistics, and distribution under one contract.
- Core Business Focus: Allows biotech teams and CDMOs to concentrate on their primary business functions.
- Minimized Touch Points: Reduces risk through combined cryopreservation, logistics, and biostorage services.
Apheresis Sourcing & Collection Partnerships
Through strategic partnerships, Cryoport Systems enhances its supply chain capabilities to provide access to highly characterized, quality-controlled, manufacture-ready cryopreserved leukopaks. Our collaborations with world-renowned organizations ensure the highest standards of donor material collection and management.
- Strategic Partnerships: Access to 7 million+ genetically diverse potential donors through strategic partnerships with NMDP BioTherapies and others.
- High-quality Collection: Established network ensures compliance and quality.
- Global Support: Enables delivery of optimized, manufacture-ready cryopreserved leukopaks across the U.S. and Europe.
Supporting certainty through our risk-mitigating supply chain expertise — one patient, one therapy, one product at a time.

Optimizing Cell Therapy with IntegriCell® Automated Cryopreservation
Learn how the IntegriCell® platform optimizes cryopreservation with automated closed processing, maintaining cell viability and recovery across multiple donor-derived leukaphereses.
Unparalleled Scope of Robust Supply Chain Management Solutions and Trusted Logistics
Cryoport Systems is Enabling the Outcome® through our unparalleled supply chain platform.
Cryoport Systems offers an integrated supply chain platform featuring temperature-controlled solutions that span the entire journey of cellular therapies. From initial collection through cryopreservation, biostorage, and final delivery, our comprehensive services ensure the highest quality and consistency of manufacture-ready cryopreserved leukopaks. Our robust global infrastructure and extensive logistics network allow us to efficiently support clients across diverse geographic regions.
Leveraging IntegriCell’s® cryopreservation services within our integrated supply chain framework provides seamless integration into BioServices and biostorage, consulting, shipping systems, and advanced logistics. This unified approach under a single vendor relationship simplifies the process, mitigates risks, and optimizes costs. Our end-to-end capabilities ensure that every step – from donor material collection to the delivery of lifesaving therapies – is managed with precision and care, supporting the successful development and administration of advanced cell therapies.
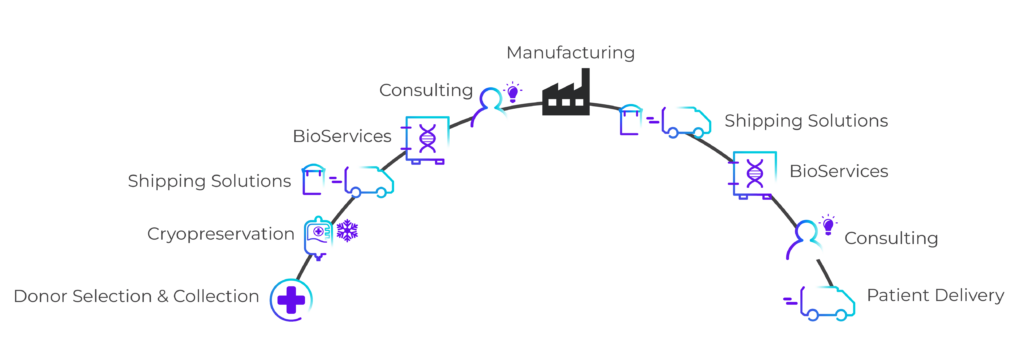
IntegriCell™ Cryopreservation
Our standardized cryopreservation protocols and utilization of innovative technologies support the biopharmaceutical supply chain from research stages to clinical operations and process development to manufacturing. Integrated into a platform of temperature-controlled logistics excellence, storage and distribution capabilities, and industry-leading cryopreservation services, IntegriCell® helps to ensure the safety, quality, and viability of critical biotherapeutic materials.
Shipping Systems
Cryoport Systems has developed and implemented the only temperature-controlled shipping systems in the industry of their kind that adhere to ISO 21973 standards and support various temperature ranges. While other companies rely on rented dewars to complete shipments, Cryoport Systems owns and operates our specially created diverse shipper system product lines, referred to as Cryoport Elite® and Cryoport Express®.
Our shipping systems undergo meticulous engineering to achieve unparalleled quality. They’re also supported by Veri-Clean®, our fully validated cleaning and disinfection process, along with our Chain of Compliance® standard, which provides end-to-end accountability. We incorporate state-of-the-art informatics and data monitoring technology into our systems to provide near real-time traceability. Clients trust us because our processes are proven to maintain both Good Manufacturing Practice (GMP) and Good Distribution Practice (GDP) standards no matter the material type.
BioServices & Biostorage
We’re proud to introduce our BioServices solutions at our Global Supply Chain Centers. These state-of-the-art facilities combine our existing logistics processes and capabilities with our new, cutting-edge BioServices infrastructure all under one roof.
We’ve designed this new and integrated approach to support cell and gene therapy manufacturing and development. This includes comprehensive temperature-controlled storage, fulfilment, kit production, secondary packaging, labelling of therapeutic products, and storage of GMP raw materials all in addition to our world-class logistics.
Consulting Services
Complex biologics and cell and gene therapies require a robust, temperature-sensitive supply chain. With our Consulting Services, you have an expert at hand who is full of crucial advice and knowledge and who can leverage decades of experience and the data from more than 800,000 successful shipments to quickly spot and solve any risks in your temperature-controlled supply chain and logistics. We tailor solutions that deliver value and insight with robust services that encompass everything from shipping risk assessments to packaging qualifications, custom packaging, shipping lane validation studies, and more. Whether you are pre-IND or post-commercial, we will work with you to tailor a solution that meets your needs, timelines, and budget.
Transportation
When it comes to shipping sensitive materials, Cryoport Systems’ transportation services can be trusted every step of the way. Our logistics capabilities meet the transportation needs of a wide range of materials for the biopharmaceutical, reproductive medicine, and animal health markets. We proudly provide the safe transportation of commodities at temperatures including cryogenic, ultra-cold (dry ice), 2-8˚C, and controlled room temperature. For assistance in moving a single sample across town or shipping an entire fleet of sensitive material from one country to another, you can rely on us and our logistics partners to transport your commodities with their integrity intact.
Additionally, our Cryoshuttle® pickup and delivery service provides local sensitive material transportation within proximal distance to all Cryoport Systems’ Global Logistics Centers and Global Supply Chain Centers. For materials that need to be shipped internationally, our team of knowledgeable experts can help coordinate customs clearance across borders. Whether it’s domestic or global sensitive material handling, we can accommodate your temperature-controlled supply chain needs.
You can TRUST our Comprehensive and Integrated Supply Chain Platform to Enable Your Outcomes
Let’s start a conversation – tell us how we can help:
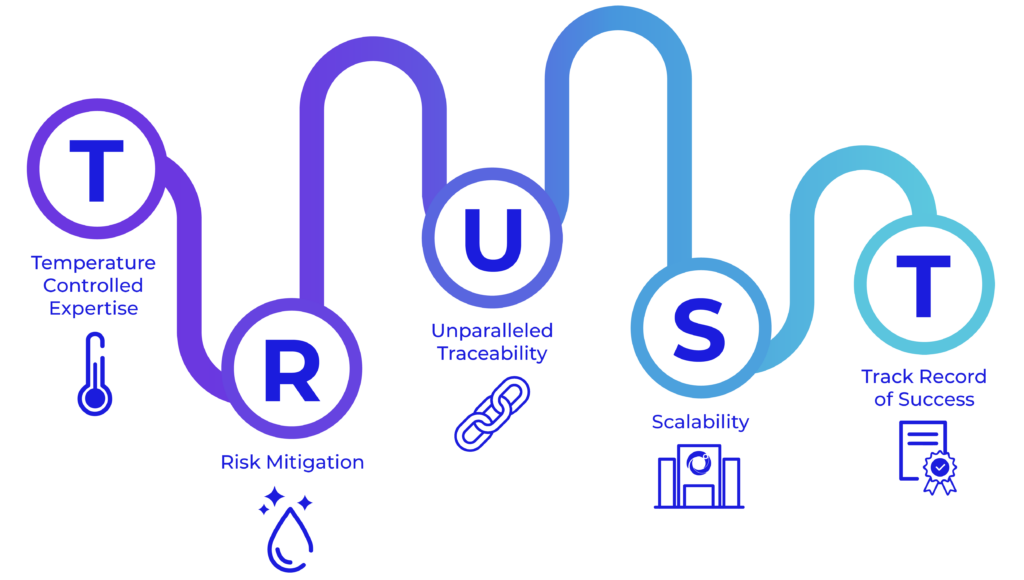
At Cryoport Systems, we are purpose-built for robust, comprehensive support with a longstanding dedication to quality.
We lead the way in temperature-controlled expertise for supply chain support with flexible solutions that meet your needs. We are the only provider in the life science industry to requalify our shippers after every single use through our proprietary Veri-Clean® process and our robust requalification procedures. Thanks to our Chain of Compliance® standard, we stand at the forefront of unparalleled traceability in managing the distribution of irreplaceable commodities. We offer scalability with expanded depth of services – such as BioServices and biostorage, consulting, and cryopreservation – that add certainty to your supply chain with tailored solutions that fit your needs. With 650+ clinical trials supported and more than half a million successful shipments of sensitive – and often irreplaceable – materials across the globe, you can count on our track record of success that spans decades.