Animal Health Archives

10/21/2025
Quality Assurance in Animal Health Supply Chains: How to Mitigate Risks
In the animal health industry, innovation doesn’t end when a new therapy leaves the lab. From the moment a product is ready to move, its quality depends on one thing above all else: a controlled, reliable supply chain. In this context, quality assurance (QA) goes beyond a regulatory requirement and quickly becomes a critical safeguard that ensures every shipment maintains its efficacy and compliance throughout the complexities of transport.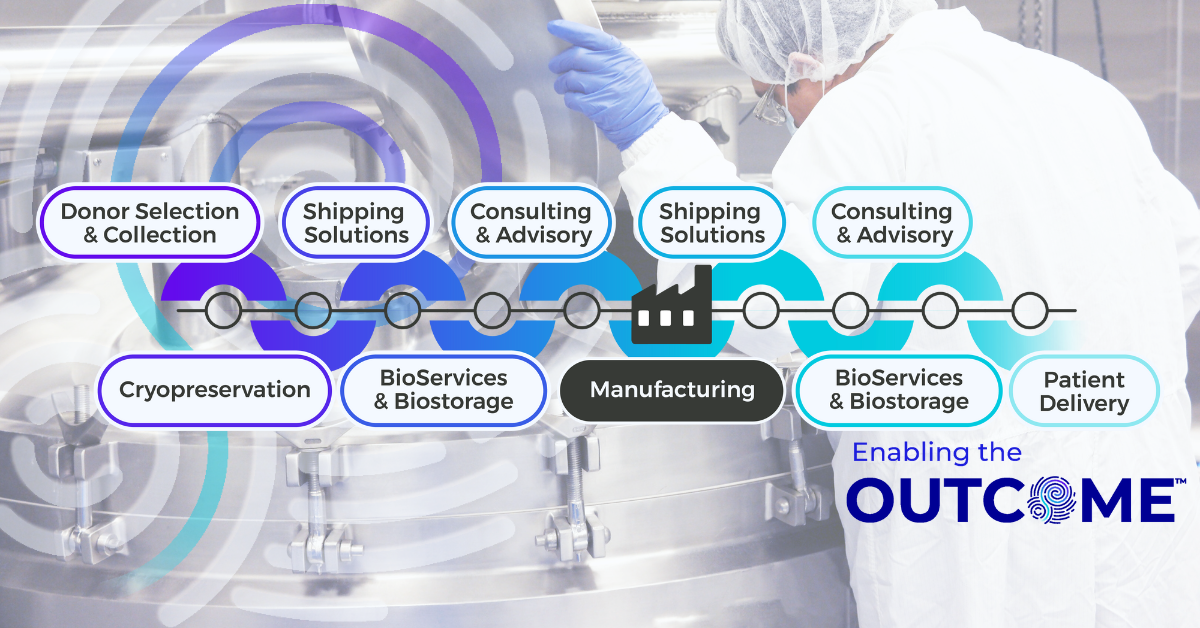
10/15/2025
The Connected Chain: Why Supply Chain Integration Matters in Advanced Therapy Manufacturing
Contract Development and Manufacturing Organizations (CDMOs) are at the heart of the advanced therapy ecosystem, managing complex, high-stakes programs that demand speed and compliance. As therapies move from early development into clinical trials and through to commercialization, every stage of manufacturing, packaging, biostorage, and final drug delivery need to align perfectly for seamless execution. Yet in many cases, these functions are managed by multiple vendors, each operating in isolation. The result is a fragmented supply chain that increases risk while slowing execution due to layers of operational burden.
09/23/2025
Revolutionizing Animal Health Logistics: How Advanced Technology Protects Product Quality
Innovation in the animal health sector extends far beyond the laboratory. While researchers develop next-generation vaccines, advanced biologics, and complex veterinary therapeutics, the logistics that bring these products from manufacturing to the field are evolving just as rapidly. For companies delivering temperature-sensitive products across continents, ensuring quality has quickly become mission-critical. A shipment that arrives late, compromised, or out of specification can affect patient outcomes and regulatory compliance, all while quietly eroding operational efficiency and profit margins.
08/01/2025
Built for Speed: How Cryoport Systems Delivers Without Compromise
In the life sciences, time isn’t just money, it impacts patient outcomes, regulatory compliance, and even trial integrity. When a temperature-sensitive shipment is delayed due to inventory shipper shortages or logistical bottlenecks, the impact can cascade across an entire program. Unfortunately, these scenarios aren’t theoretical. When you’re working with irreplaceable biologic materials, this kind of disruption quickly moves from inconvenient to unacceptable.
06/17/2025
Ensuring Safe, On-Time Deliveries for Animal Biopharma
Transporting advanced therapeutics for the animal health industry is just as time- and temperature-sensitive as human therapeutics, yet it is not often managed with the same level of care. This results in biologics and vaccines running into preventable risk during transit. Whether you’re supporting a clinical trial, delivering commercial supply, or responding to a health emergency, ensuring the quality, compliance, and visibility of every shipment is crucial.
04/16/2025
Delivering Over 5 Billion Doses of Biotherapeutics to Protect Animal Health Worldwide
In 2024, Cryoport Systems achieved a remarkable milestone: the delivery of over 5 billion doses of biotherapeutics, including cell-based therapies, antibody-based therapies, and vaccines to the global animal health industry. This achievement is the result of our unwavering commitment to ensuring the health and well-being of animals across the world, from livestock that sustain global food supply chains to the beloved companion animals that enrich our lives.
03/24/2025
Strengthening Global ATMP Logistics: How Cryoport Systems’ Netherlands Facility Supports Temperature-Controlled Supply Chains
The success of ATMPs depends on more than just scientific breakthroughs, it requires a highly controlled, risk-mitigated supply chain that ensures product integrity from research through commercialization. With cell and gene therapies moving across multiple clinical sites, manufacturing facilities, and regulatory jurisdictions, developers need a logistics partner that can support seamless, temperature-controlled supply chain management at a global scale.
03/12/2025
The Gateway to Europe: How Cryoport Systems’ Stevenage Supply Chain Hub Supports ATMP Development
For UK-based Advanced Therapy Medicinal Product (ATMP) developers, access to European clinical trial sites and commercial markets is a critical component of a successful growth strategy. Yet, navigating regulatory barriers, customs challenges, and supply chain complexities can create unnecessary delays, increase costs, and put the integrity of sensitive biologics at risk.
12/30/2024
Supporting Compliance and Reducing Risk in Gene Therapy Logistics
When it comes to high stakes logistics in the gene therapy sector, adherence to regulatory standards isn’t just a box to check—it’s essential to delivering therapies that maintain integrity from production to patient. Cryoport Systems understands the unique risks in this space and has built compliance and risk mitigation into the very design of the Cryoport Elite™ Ultra Cold shipping system.Categories
- All (48)
- Animal Pharmaceuticals (18)
- Expert Insights (12)
- Livestock Management (10)
- Veterinary Support (14)
